Changing the course of severe rare diseases
A new approach
for patients suffering from ADPKD
SCY-770: A well-characterized, first-in-class, clinical stage, selective direct AMPK activator, under development as a differentiated, disease-modifying therapy for patients with Autosomal Dominant Polycystic Kidney Disease (ADPKD)
ADPKD is a progressive, inherited condition in which fluid filled cysts develop and enlarge in the kidneys over time. As the disease advances, kidney function can decline, and many people ultimately face kidney failure that may require dialysis or transplant. We are advancing a disease modifying approach based on direct activation of AMPK, a key regulator of cellular energy pathways implicated in cyst growth. This novel MOA is supported by a compelling mechanistic rationale in ADPKD.
Direct Allosteric Activation of AMPK by SCY-770
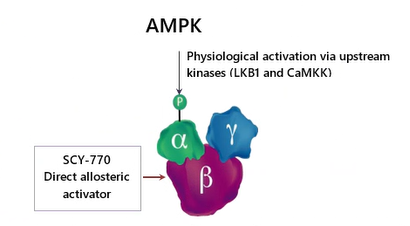
Directly activating AMPK SCY-770 has the potential to counteract multiple mechanisms simultaneously, meaningfully altering disease progression for patients facing very limited treatment options.
Key effects of AMPK activation in ADPKD:
- Inhibits mTOR signaling - reduces pathological cell proliferation
- Reduces CFTR-mediated chloride transport - decreases cyst fluid secretion
- Suppresses cAMP signaling pathways
- Reduces aerobic glycolysis (Warburg effect)
- Decreases inflammation and fibrosis

Clinical Stage:
- SCY-770 is an orally administered product with an advanced clinical development program, including 8 completed clinical trials with a favorable safety profile. Studies in patients with ADPKD are under planning.
Preclinical Validation:
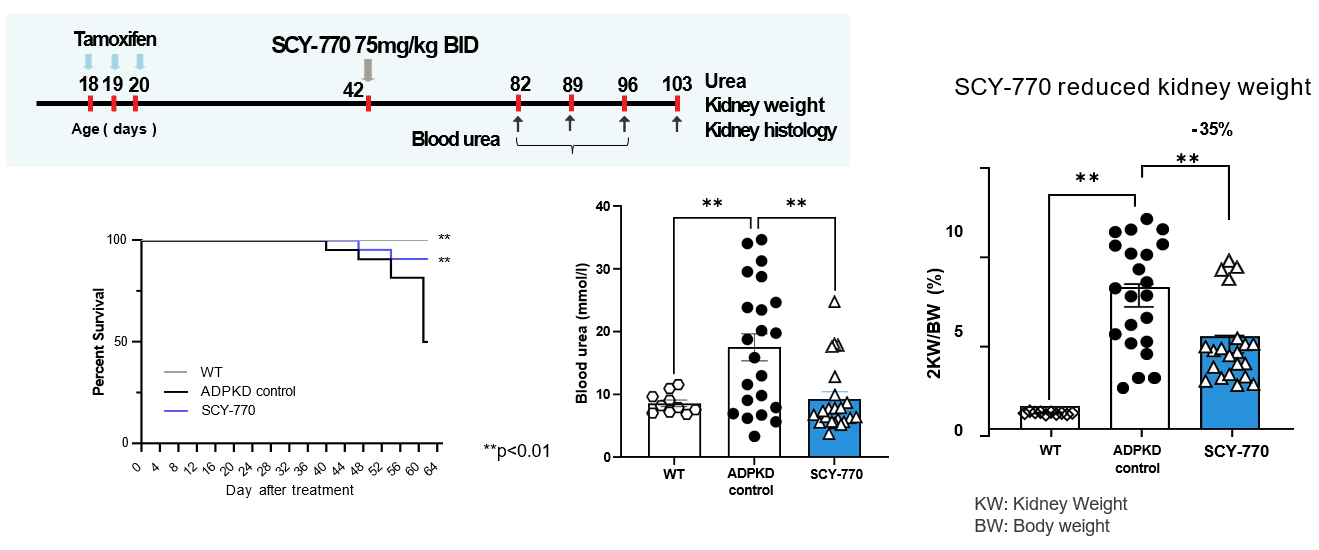
Robust in vivo Efficacy In preclinical ADPKD models, SCY-770 demonstrated:
- Reduced kidney weight (cyst growth)
- Improved renal function (blood urea)
- Significant survival benefit vs control
- Reduced inflammation
These findings support the potential for meaningful clinical impact and long-term disease control.
To learn more about the effect of SCY-770 in ADPKD mice model: DOI: 10.1016/j.kint.2023.01.026